

Certified Researcher – RCI (Verified Credential- RAI-2026-466151)
Credentialed in research methodology, documentation integrity, and compound handling standards. Scan the QR code to verify authenticity and enter gold numbers into site when you get transferred to page!
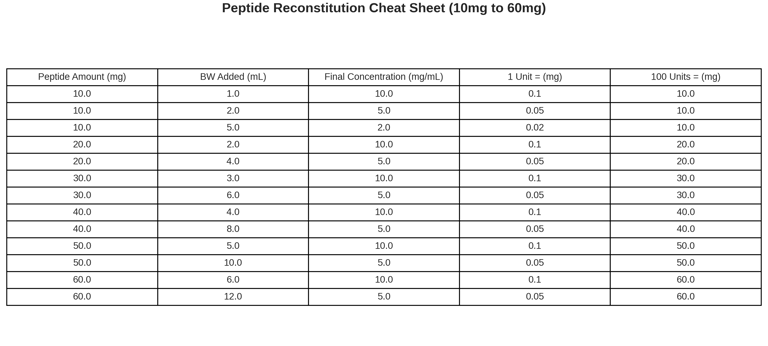
Reconstitution Cheat Sheet (Research-Only)

METABOLIC & GLP-RELATED RESEARCH
Below are peptides commonly studied in metabolic, appetite, and glucose-related research models.
• Semaglutide- A synthetic peptide analog of the GLP‑1 hormone, studied for its role in metabolic signaling and glucose regulation. It binds to GLP‑1 receptors involved in insulin secretion, gastric‑emptying pathways, and appetite‑related neurocircuitry. Researchers use Semaglutide to investigate how enhanced GLP‑1 receptor activation influences metabolic homeostasis, nutrient processing, and energy‑balance mechanisms.
• Tirzepatide-A synthetic peptide investigated for its ability to modulate multiple metabolic signaling pathways. It acts as a dual agonist at the GIP and GLP‑1 receptors, allowing researchers to study how simultaneous activation influences insulin signaling, glucose regulation, and energy‑balance mechanisms. Current research explores its effects on pancreatic function, appetite‑related neurocircuitry, and metabolic homeostasis.
• Retatrutide- A next‑generation tri‑agonist peptide designed to activate GIP, GLP‑1, and glucagon receptors. Researchers use it to examine how multi‑pathway stimulation affects energy expenditure, glucose metabolism, and lipid utilization. Its unique receptor profile makes it a valuable tool for studying integrated metabolic control and whole‑body energy regulation.
• Cagrilintide- A long‑acting amylin analogue used in research to investigate satiety signaling and gastric‑emptying pathways. It interacts with amylin receptors in the brainstem and hypothalamus, helping researchers better understand appetite regulation, nutrient sensing, and the coordination of digestive processes.
• AOD-9604- A fragment of the human growth hormone molecule (specifically the 176–191 region) studied for its role in lipid metabolism. It does not activate GH receptors but instead is used to explore how this specific peptide segment influences fat‑cell signaling, energy utilization, and metabolic pathways independent of growth hormone activity.
• MOTS-c- A mitochondrial‑derived peptide encoded within mitochondrial DNA. It is researched for its role in cellular energy regulation, AMPK activation, and metabolic stress responses. Studies focus on how MOTS‑c influences glucose utilization, exercise‑related adaptations, and mitochondrial communication with the nucleus.
RECOVERY & TISSUE-RESPONSE RESEARCH
Recovery & Tissue Response
These peptides are often studied in models involving cellular response, tissue repair, and recovery pathways.
• BPC-157- A synthetic peptide fragment derived from the naturally occurring Body Protection Compound found in gastric tissue. It is studied for its influence on angiogenesis, cellular repair pathways, and inflammatory modulation. Researchers use BPC‑157 to explore how it interacts with growth‑factor signaling, tissue regeneration processes, and gastrointestinal protective mechanisms.
• TB-500- A synthetic version of a segment of thymosin beta‑4, a protein involved in cellular structure and movement. TB‑500 is used in research to investigate actin regulation, cell migration, and tissue‑repair pathways. Studies often focus on its role in angiogenesis, wound‑healing mechanisms, and cytoskeletal organization.
• BPC-157 + TB-500- A research combination pairing two peptides studied for their complementary roles in cellular repair and angiogenesis. This blend allows researchers to examine how simultaneous modulation of growth‑factor signaling, inflammatory pathways, and actin‑related cell migration may influence tissue‑recovery processes. It is commonly used to explore synergistic effects on regenerative biology.
• GHK-Cu- A naturally occurring copper‑binding peptide found in human plasma and tissues. GHK‑Cu is studied for its role in skin biology, collagen synthesis, and cellular remodeling. Research focuses on its interactions with gene expression related to repair, antioxidant activity, and tissue regeneration, as well as its ability to deliver copper ions to cells.
• GLOW- A multi‑peptide research blend formulated to investigate pathways related to cellular repair, skin health, and metabolic signaling. Its components are selected to allow researchers to study combined effects on collagen support, tissue regeneration, and systemic metabolic pathways. GLOW is used to explore how multi‑pathway peptide interactions may influence overall cellular vitality.
• KLOW- A comprehensive research formulation combining peptides associated with regenerative signaling, metabolic modulation, and anti‑inflammatory pathways. KLOW is designed to help researchers examine how multi‑peptide synergy affects tissue repair, angiogenesis, and cellular communication. Its broad profile makes it useful for studying complex biological interactions across multiple systems.
NEUROLOGICAL & COGNITIVE PATHWAYS
Neurological & Cognitive
These compounds are studied in research involving cognitive pathways, neurological signaling, and receptor activity.
• Selank- A synthetic peptide analog derived from the endogenous tuftsin molecule. Selank is studied for its influence on neuropeptide signaling, particularly pathways related to stress response, cognitive processes, and neurotransmitter modulation. Researchers use it to explore how it affects GABAergic activity, memory formation, and central nervous system regulation.
• Semax- A synthetic fragment of the ACTH(4‑10) peptide modified for enhanced stability. Semax is researched for its effects on neurotrophic signaling, cognitive pathways, and stress‑response mechanisms. Studies focus on its interaction with BDNF‑related processes, attention regulation, and neuroprotective molecular cascades.
• PT-141- A synthetic peptide analog that targets melanocortin receptors, particularly MC3R and MC4R. PT‑141 is used in research to investigate neural pathways involved in arousal signaling, reward circuits, and hypothalamic regulation. Its receptor activity makes it valuable for studying melanocortin‑mediated central nervous system processes.
GROWTH HORMONE PATHWAYS
Growth Hormone Pathways
These peptides are used in research involving GH-related signaling and receptor interactions.
• Sermorelin- A synthetic analog of growth hormone–releasing hormone (GHRH) used in research to study pituitary signaling and growth‑hormone regulatory pathways. Researchers examine Sermorelin to better understand hypothalamic–pituitary communication, pulsatile GH release, and downstream metabolic and cellular effects associated with GH modulation.
• Ipamorelin- A selective ghrelin‑receptor (GHSR) agonist studied for its role in stimulating growth‑hormone secretion. Research focuses on its high receptor specificity, minimal off‑target activity, and its influence on pituitary signaling, appetite‑related pathways, and neuroendocrine regulation.
• CJC-1295 + Ipamorelin- A research combination pairing a GHRH analog (CJC‑1295) with a selective ghrelin‑receptor agonist (Ipamorelin). This blend allows researchers to study how dual‑pathway stimulation—GHRH and GHSR—affects pulsatile GH release, pituitary signaling, and broader endocrine regulatory mechanisms. It is commonly used to explore synergistic effects on growth‑hormone dynamics.
• Tesamorelin- A stabilized analog of growth hormone–releasing hormone (GHRH) designed for extended activity. Tesamorelin is used in research to investigate pituitary GH secretion, IGF‑1–related pathways, and metabolic signaling. Studies often focus on its enhanced stability and its ability to model long‑acting GHRH stimulation.
NAD+
NAD+ Research
NAD+ is commonly used in studies involving cellular energy, mitochondrial function, and redox pathways.
• NAD+ - A naturally occurring coenzyme present in all living cells, essential for redox reactions and cellular‑energy metabolism. NAD+ is studied for its role in mitochondrial function, electron‑transport activity, and regulation of sirtuin pathways. Researchers use it to investigate how shifts in NAD+/NADH balance influence metabolic signaling, DNA‑repair processes, and overall cellular homeostasis.








